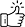Development of Sustainable Technologies Using Computational and Synthetic Tools
Chemistry is the most powerful field which can help provide immediate and promising solutions to global warming and climate change by designing and developing technologies for a sustainable future, writes Vidya Avasare

Vidya Avasare
8 June, 2023 | 4m readContinuous carbon emission in the atmosphere is contributing to global warming and climate change; which is adversely affecting life on the earth. If carbon emission continues to increase then there would be no life on the earth! Therefore, it is crucial to address carbon emissions by accelerating fundamental as well as advanced research. Among all, chemistry would be the most powerful field which would help to provide immediate and promising solutions by designing and developing sustainable technologies for a sustainable future as follows:
- Carbon Dioxide Capture: Capturing of atmospheric CO2 at the source (oil refineries, mines, industries, etc.).
- Carbon Dioxide Utilization: Convert captured CO2 to fuels and value-added bulk chemicals.
- Green Hydrogen Production: Hydrogen is a green fuel and it is also needed for CO2 utilization.
- Green Chemistry Approach: It is important to replace existing environmentally hazardous and expensive technologies with inexpensive and environmentally benign ones to reduce CO2 emissions and other pollutants.
Experimental catalyst discovery is a dynamic process and requires the number of multiple parameters, resources, and time to develop robust catalysts for various applications (Figure 1). Recently, computational catalysis has been considered one of the promising methods in developing energy-efficient and inexpensive technologies and for the screening and designing of catalysts for a wide range of applications. Further, it is also advantageous to develop machine learning models to test and demonstrate the catalytic performance of millions of catalysts in shorter times. We have been using density functional theoretic (DFT) study to identify efficient and inexpensive carbon dioxide to C1 as well as multiple carbon products.
We are particularly interested in methanol production as it has many-fold benefits. Methanol can be used as fuel and chemical hydrogen storage material, and it is also used as either a solvent or precursor for most commercial products. However, to utilize carbon dioxide, hydrogen is required, and hence sustainable and inexpensive production of hydrogen is also necessary and hydrogen can be produced from alcohol or water. Thus, carbon sequestration and hydrogen productions are important steps of the circular carbon economy. (What is the circular economy – and why is the world less circular? | World Economic Forum (weforum.org)).
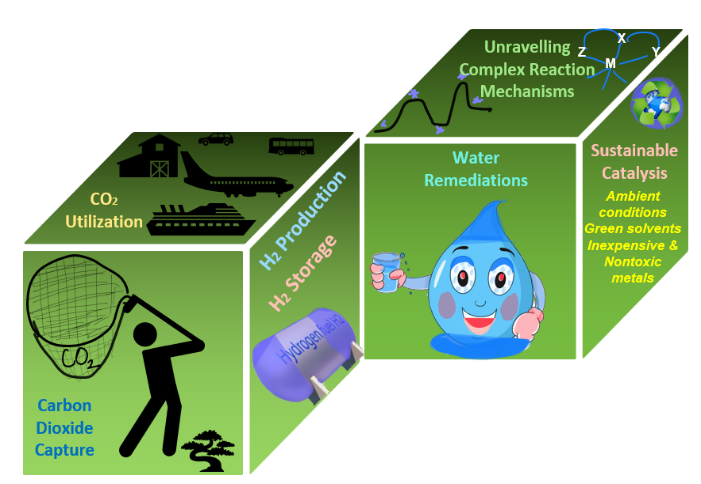
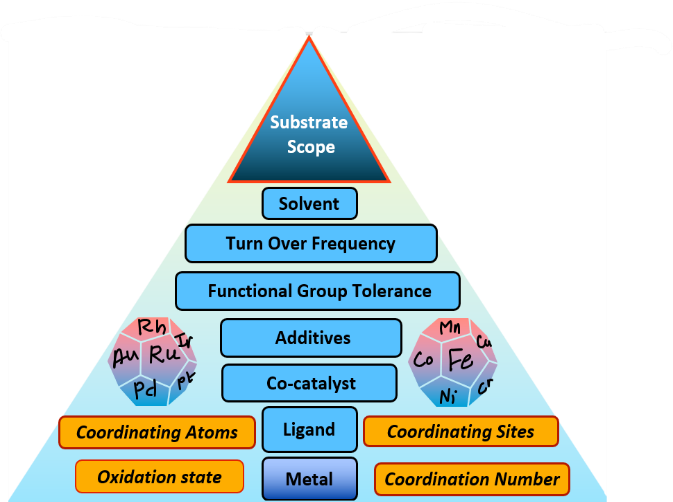
Recently, we received a research grant from SERB under the CRG scheme (File no. CRG/2022/003758) for computational designing and development of first-row 3d transition metal catalysts to produce H2 from methanol. Apart from CO2 utilization and H2 production, we are also working on developing carbon capture materials and porous organic polymers for water remediation using DFT methods under the Stars project scheme (Stars (iisc.ac.in)) in collaboration with Professor Sujit Ghosh, IISER Pune (Figure 2). The computationally predicted lead catalysts are used to demonstrate their catalytic performance using experimental methods and investigating inexpensive and environmentally benign homogeneous and heterogeneous catalysts to demonstrate C—X (X = C, N, O) coupling reactions and C—H activation reactions under ambient conditions (Figure 2). Many of these synthetic protocols are useful in developing small organic molecules for various applications in medicines, materials, diagnostics, etc. without compromising sustainability.
(The writer is a Visiting Professor in the Department of Chemistry at Ashoka University)